 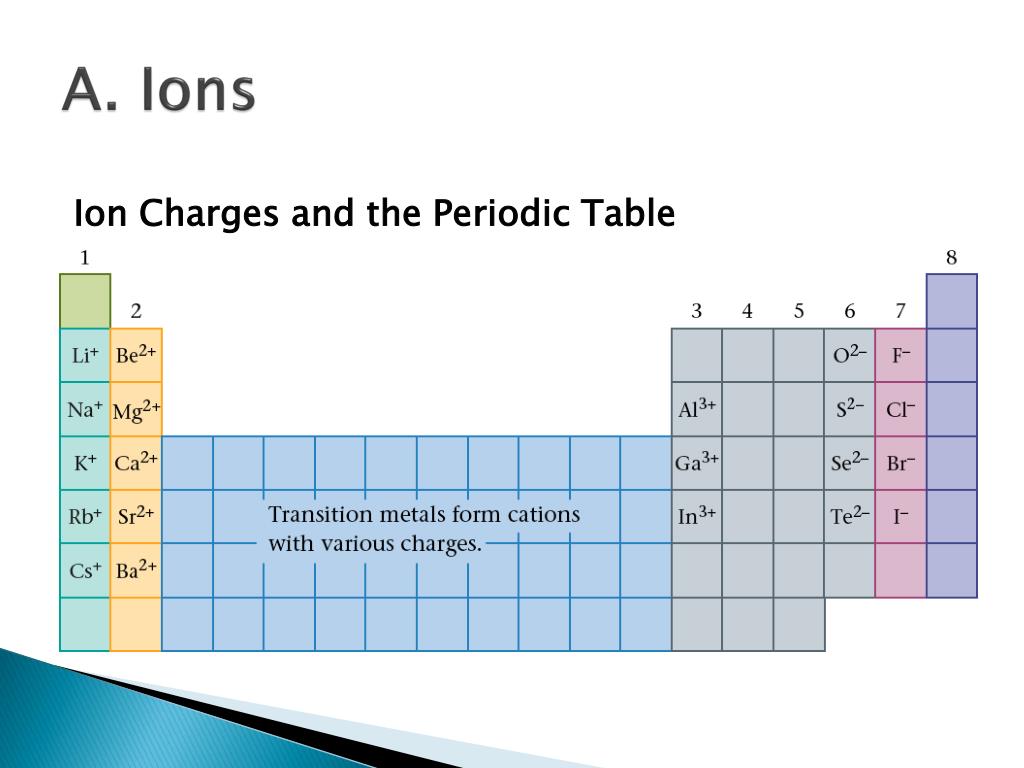
In these lower energy states, the outermost energy level has eight electrons (an “octet”). This instability drives them toward the lower energy states represented by the noble gases that are nearby in the periodic table. The elements in the other groups have subshells that are not full, so they are unstable when compared to the noble gases. They are already at a low energy state, so they tend to stay as they are. These elements have electron configurations characterized by full s and p subshells. For atoms, these lower energy states are represented by the noble gas elements. Lower energy configurations are more stable, so things are naturally drawn toward them. Throughout nature, things that are high in energy tend to move toward lower energy states. Deviations from this ratio result in charged particles called ions. This one-to-one ratio of charges is not, however, the most common state for many elements. The overall charge on the atom is zero, because the magnitude of the negative charge is the same as the magnitude of the positive charge. This is because the number of electrons (negative in charge) is equal to the number of protons (positive in charge). 
Up until now we have been discussing only the elemental forms of atoms which are neutrally charged. 3.1 Introduction to the Octet Rule 3.2 Ions and the Periodic Table Common Cations Common Anions Ions of Transition Metals 3.3 Ionic Bonding 3.4 Practice Writing Correct Ionic Formulas 3.5 Naming Ions and Ionic Compounds 3.6 Polyatomic Ions 3.7 Naming Polyatomic Ions 3.8 Properties and Types of Ionic Compounds 3.9 Arrhenius Acids and Bases 3.10 Focus on the Environment – Acid Rain 3.11 Chapter Summary 3.12 References This text is published under creative commons licensing, for referencing and adaptation, please click here. CH104: Chapter 3 – Ions and Ionic Compounds 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
